Abstract
Objective:
To determine the half-life of serum caffeine concentrations and its relation to apnea of prematurity (AOP) after caffeine is discontinued in preparation for hospital discharge.
Study Design:
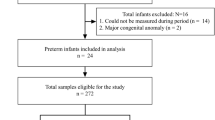
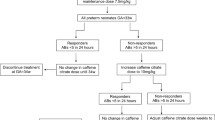
Prospective cohort study involving preterm infants with gestational ages ⩽33 weeks at birth. After caffeine was discontinued, serum caffeine concentrations and electronic detection of pathologic apnea, defined a priori, were obtained at 24 and 168 h, respectively.
Result:
Caffeine levels decreased from 13.3±3.8 to 4.3±2 mg l−1 (n=50, mean±s.d.) at 24 and 168 h, respectively (P<0.01). The mean caffeine half-life was 87±25 h at 35±1 weeks postmenstrual age. Seven days after discontinuation of caffeine, 64% of the infants had pathologic apnea.
Conclusion:
Hospital discharge planning for preterm infants with a history of AOP should be carefully considered after discontinuing caffeine. This study showed that caffeine may not reach subtherapeutic levels until around 11–12 days.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lodha A, Seshia M, McMillan DD, Barrington K, Yang J, Lee SK et al. Association of early caffeine administration and neonatal outcomes in very preterm neonates. JAMA Pediatr 2015; 169 (1): 33–38.
Schmidt B, Roberts RS, Davis P, Doyle LW, Barrington KJ, Ohlsson A et al. Caffeine therapy for apnea of prematurity. N Engl J Med 2006; 354 (20): 2112–2121.
Eichenwald EC, Aina A, Stark AR . Apnea frequently persists beyond term gestation in infants delivered at 24 to 28 weeks. Pediatrics 1997; 100 (3 Pt 1): 354–359.
Merritt TA, Pillers D, Prows SL . Early NICU discharge of very low birth weight infants: a critical review and analysis. Semin Neonatol 2003; 8 (2): 95–115.
Ramanathan R, Corwin MJ, Hunt CE, Lister G, Tinsley LR, Baird T et al. Cardiorespiratory events recorded on home monitors: comparison of healthy infants with those at increased risk for SIDS. JAMA 2001; 285 (17): 2199–2207.
Charles BG, Townsend SR, Steer PA, Flenady VJ, Gray PH, Shearman A . Caffeine citrate treatment for extremely premature infants with apnea: population pharmacokinetics, absolute bioavailability, and implications for therapeutic drug monitoring. Ther Drug Monit 2008; 30 (6): 709–716.
Natarajan G, Botica ML, Thomas R, Aranda JV . Therapeutic drug monitoring for caffeine in preterm neonates: an unnecessary exercise? Pediatrics 2007; 119 (5): 936–940.
Leon AE, Michienzi K, Ma CX, Hutchison AA . Serum caffeine concentrations in preterm neonates. Am J Perinatol 2007; 24 (1): 39–47.
Winter ME . Basic Clinical Pharmacokinetics. Lippincott Williams & Wilkins: Philadelphia, 2004.
Darnall RA, Kattwinkel J, Nattie C, Robinson M . Margin of safety for discharge after apnea in preterm infants. Pediatrics 1997; 100 (5): 795–801.
Aranda JV, Beharry K, Valencia GB, Natarajan G, Davis J . Caffeine impact on neonatal morbidities. J Matern Fetal Neonatal Med 2010; 23 (Suppl 3): 20–23.
Davis PG, Schmidt B, Roberts RS, Doyle LW, Asztalos E, Haslam R et al. Caffeine for apnea of prematurity trial: benefits may vary in subgroups. J Pediatr 2010; 156 (3): 382–387.
Le Guennec JC, Billon B, Pare C . Maturational changes of caffeine concentrations and disposition in infancy during maintenance therapy for apnea of prematurity: influence of gestational age, hepatic disease, and breast-feeding. Pediatrics 1985; 76 (5): 834–840.
Alur P, Bollampalli V, Bell T, Hussain N, Liss J . Serum caffeine concentrations and short-term outcomes in premature infants of 29 weeks of gestation. J Perinatol 2015; 35: 434–438.
Comer AM, Perry CM, Figgitt DP . Caffeine citrate: a review of its use in apnoea of prematurity. Paediatr Drugs 2001; 3 (1): 61–79.
Aldridge A, Aranda JV, Neima AH . Caffeine metabolism in the newborn. Clin Pharmacol Ther 1979; 4: 447–453.
Amin SB, Burnell E . Monitoring apnea of prematurity: validity of nursing documentation and bedside cardiorespiratory monitor. Am J Perinatol 2013; 30 (8): 643–648.
Di Fiore JM, Arko MK, Miller MJ, Krauss A, Betkerur A, Zadell A et al. Cardiorespiratory events in preterm infants referred for apnea monitoring studies. Pediatrics 2001; 108 (6): 1304–1308.
Subhani M, Katz S, DeCristofaro JD . Prediction of postdischarge complications by predischarge event recordings in infants with apnea of prematurity. J Perinatol 2000; 20 (2): 92–95.
Ho T, Dukhovny D, Zupancic JAF, Goldmann DA, Horbar JD, Pursley DM . Choosing wisely in newborn medicine: five opportunities to increase value. Pediatrics 2015; 136 (2): e482–e489.
Haddad W, Sajous C, Hummel P, Guo R . Discontinuing caffeine in preterm infants at 33-35 weeks corrected gestational age: failure rate and predictive factors. J Neonatal Perinatal Med 2015; 8 (1): 41–45.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Doyle, J., Davidson, D., Katz, S. et al. Apnea of prematurity and caffeine pharmacokinetics: potential impact on hospital discharge. J Perinatol 36, 141–144 (2016). https://doi.org/10.1038/jp.2015.167
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2015.167
This article is cited by
-
Longitudinal pilot study of oxygen saturation indices in healthy preterm infants
Pediatric Research (2023)
-
Variations in length of stay among survived very preterm infants admitted to Chinese neonatal intensive care units
World Journal of Pediatrics (2022)
-
Immature control of breathing and apnea of prematurity: the known and unknown
Journal of Perinatology (2021)